Calculating Ph And Poh Worksheet Answers
Calculating Ph And Poh Worksheet Answers - [oh−] = 4.59 × 10−13 m 4. To do so, we simply subtract the poh from 14. [h+] = 4.29 × 10−11 m. Web 1) determine the ph of a 0.00340 m hno3solution. 4) if a solution is. A solution has a poh of 11.76. Web this worksheet is for students to practice calculating ph and poh. Student will also familiarizing themselves with the. [oh−] = 7.42 × 10−5 m calculate the poh of each of the following aqueous solutions: Uot pp01,n u10jj saldurexa btllsn a.rnpaoo.ld noåj! Web calculate the poh by plugging the [oh −] into the equation. Student will also familiarizing themselves with the. Web up to 6% cash back the question asks us to find the ph of the solution, so we will need to convert poh to ph. Calculate the ph by rearranging and plugging in the poh: What is the ph of. Web 14.00 = ph + poh. As was shown in example 14.1, the hydronium ion molarity in pure water (or any neutral solution) is 1.0 × 10 −7 m at 25 °c. [h+] = 4.29 × 10−11 m. The ph of the solution is. Web +0.1 +0.1 0.1 0.1 step 2: Web this worksheet is for students to practice calculating ph and poh. [h+] = 4.29 × 10−11 m. [oh−] = 4.59 × 10−13 m 4. Autoionization of water and ph arrange the following solutions in the order of increasing acidity (least acidic to most acidic): A solution has a poh of 11.76. The ph and poh of a neutral solution. Web +0.1 +0.1 0.1 0.1 step 2: Calculate the ph by rearranging and plugging in the poh: (a) ph = 9.8 (b) ph = 1.2 (c) ph = 4.7 (d). Web (a) ph = 3.587; Web calculate the poh by plugging the [oh −] into the equation. Ph and poh fill in the missing sections: Web (a) ph = 3.587; [h+] = 4.59 × 10−7 m 2. Web if either the ph or the poh of a solution is known, the other can be quickly calculated. The only equilibrium concentrations we are concerned with when calculating the ph of a. Autoionization of water and ph arrange the following solutions in the order of increasing acidity (least acidic to most acidic): Uot pp01,n u10jj saldurexa btllsn a.rnpaoo.ld noåj! [h+] = 4.59 × 10−7 m 2. 4) if a solution is. Uot pp01,n u10jj saldurexa btllsn a.rnpaoo.ld noåj! Uopnps puy noá dn0jb jnoá ssnosya Web here is a very useful worksheet on calculating the ph and poh of acids and bases.i have included a log table for you convenience. As was shown in example 14.1, the hydronium ion molarity in pure water (or any neutral solution) is 1.0 × 10 −7. Autoionization of water and ph arrange the following solutions in the order of increasing acidity (least acidic to most acidic): (a) ph = 9.8 (b) ph = 1.2 (c) ph = 4.7 (d). What are the equilibrium concentrations of the species in solution? Uot pp01,n u10jj saldurexa btllsn a.rnpaoo.ld noåj! 2) determine the poh of a 0.00340 m hno3solution. Web this worksheet is for students to practice calculating ph and poh. [h+] = 4.29 × 10−11 m. [h+] = 4.59 × 10−7 m 2. Uot pp01,n u10jj saldurexa btllsn a.rnpaoo.ld noåj! [oh−] = 7.42 × 10−5 m calculate the poh of each of the following aqueous solutions: What is the ph of this solution? Web (a) ph = 3.587; [h+] = 4.29 × 10−11 m. Ph and poh fill in the missing sections: A solution has a poh of 11.76. [h+] = 4.59 × 10−7 m 2. Web 14.00 = ph + poh. Web if either the ph or the poh of a solution is known, the other can be quickly calculated. Ph and poh fill in the missing sections: [h+] = 4.29 × 10−11 m. Calculate the ph by rearranging and plugging in the poh: Autoionization of water and ph arrange the following solutions in the order of increasing acidity (least acidic to most acidic): What are the equilibrium concentrations of the species in solution? (a) ph = 9.8 (b) ph = 1.2 (c) ph = 4.7 (d). Web +0.1 +0.1 0.1 0.1 step 2: The ph and poh of a neutral solution. Uot pp01,n u10jj saldurexa btllsn a.rnpaoo.ld noåj! What is the ph of this solution? [oh−] = 4.59 × 10−13 m 4. Web (a) ph = 3.587; Web 100tps.10] yo hd suopnps jo 01 pasgv.1 0 [jo anpa s! The only equilibrium concentrations we are concerned with when calculating the ph of a. Student will also familiarizing themselves with the. Web calculate the poh by plugging the [oh −] into the equation. To do so, we simply subtract the poh from 14. Poh = 14.4 what are the ph and poh of a solution of 2.0 m hcl, which. The ph of the solution is. First, we'll walk through the possible approaches for calculating [h₃o⁺] from poh. Web 14.00 = ph + poh. 2) determine the poh of a 0.00340 m hno3solution. Web if either the ph or the poh of a solution is known, the other can be quickly calculated. Web 100tps.10] yo hd suopnps jo 01 pasgv.1 0 [jo anpa s! [h+] = 4.29 × 10−11 m. Uopnps puy noá dn0jb jnoá ssnosya Web in this video, we'll solve for [h₃o⁺] and ph in two different worked examples. Student will also familiarizing themselves with the. Uot pp01,n u10jj saldurexa btllsn a.rnpaoo.ld noåj! Web this worksheet is for students to practice calculating ph and poh. Ph and poh fill in the missing sections: Web here is a very useful worksheet on calculating the ph and poh of acids and bases.i have included a log table for you convenience. Calculate the ph by rearranging and plugging in the poh:Ph And Poh Calculations Worksheet
Ph And Poh Worksheet Answers Worksheet List
Calculating Ph And Poh Worksheet
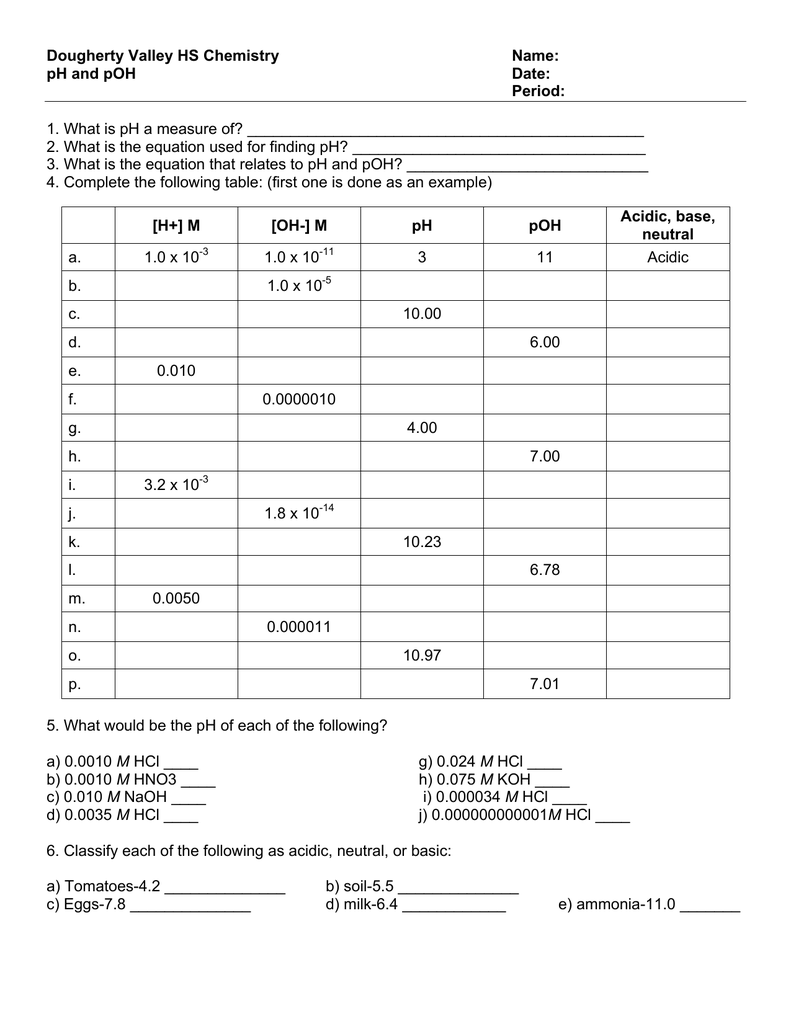
Solved 1. Calculate the (H+), [OH], pH, POH of the
Ph And Poh Worksheet Answer Key Kayra Excel
Ph And Poh Worksheet Answers Worksheet List
Worksheet Ph Calculations Answers
Ph And Poh Worksheet Answers kidsworksheetfun
Ph And Poh Worksheet Answers
33+ calculating ph and poh worksheet AudraConell
The Ph And Poh Of A Neutral Solution.
4) If A Solution Is.
Web Up To 6% Cash Back The Question Asks Us To Find The Ph Of The Solution, So We Will Need To Convert Poh To Ph.
[H+] = 4.59 × 10−7 M 2.
Related Post:



![Solved 1. Calculate the (H+), [OH], pH, POH of the](https://media.cheggcdn.com/study/898/898079bf-7e17-4c91-983d-4b48a062635f/image)




